Research
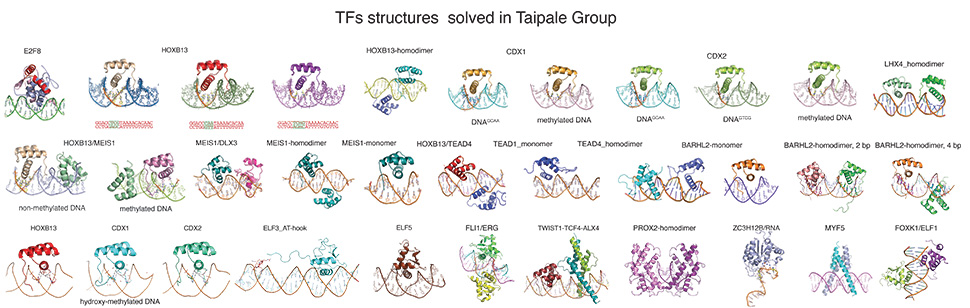
The main scientific questions addressed in our laboratory relate to the understanding of molecular mechanisms that control gene regulation through the use of high-throughput biology to characterize transcription factor binding specificities and sites in human cancer cells. TFs are analyzed alone, and in combination with other TFs and scaffolding proteins such as the mediator complex. The resulting knowledge is then applied to the interpretation of large data sets such as whole cancer genomes, and genome-wide association studies that have revealed genomic regions associated with a wide variety of diseases, including heart disease, diabetes and different types of cancer. We also develop novel and better methods such as HT-SELEX, ATI, and tools for genome editing, to carry out our work in high throughput format. The work in the laboratory is interdisciplinary, and has an impact on basic scientific understanding of gene regulation, as well as to mechanisms of formation of cancer and other diseases.
The specific objectives of our research are the following:
1) To identify mechanisms that govern transcription factor binding in vitro and in live cells
2) To use the resulting information in the interpretation of cancer genomes and genome wide-association studies
3) To validate the findings in mouse genetic models
About the laboratory
Professor Jussi Taipale got his Ph.D. from the University of Helsinki in 1996, and continued with postdoctoral work at the University of Helsinki and at Johns Hopkins University (Baltimore, MD, USA). He has headed an independent research laboratory since 2003, focusing on systems biology of growth control and cancer. The main expertise of the Taipale group is high-throughput screening and computational and experimental methods to identify causative regulatory mutations in non-protein coding DNA and to analyze genetic networks. In addition, the Taipale group has extensive expertise on mouse models of gene and regulatory region function. The group is located in three countries: we are at the University of Helsinki, Finland, Karolinska Institutet, Sweden and the University of Cambridge, UK. Currently we have seven senior scientists, three postdoctoral fellows, three graduate students, three lab managers and a personal assistant.
Highlights: